Ensuring Quality for Medical Device Displays
Medical devices—ranging from those for consumer personal use to those designed for clinical settings—must adhere to stringent requirements in their design and manufacture. Performance, accuracy, and reliability are vital when patient health is on the line. The increasing digitization of medical practice and care management has led to a boom in integrated medical devices. The IoT-enabled medical device market is forecast to grow to $63.43 billion by 2023 at a rate of 25.2% CAGR.1 Smart devices that are “more automated and connected are improving the state of healthcare, making it possible to perform remote surgeries with doctors on the other side of the globe.”2
Within the medical device market, the use of wearables is also expanding rapidly. Encompassing sports and fitness trackers, patient remote monitoring, and home healthcare devices (for both diagnostic and therapeutic purposes), the wearable medical device market is expected to grow by more than 17% CAGR, reaching $29 billion in global revenues by 2026.3

Examples of medical device displays in a hospital room (left), a blood sugar monitor + sensor for patient use (center), and the passive segmented display on a blood pressure monitor (right). (Left and right images: New Vision Display, center image: FreeStyle Libre)
Driving this growth in medical devices are several factors:
- Increasing incidence of health conditions – Aging population demographics and the growing incidence of chronic diseases like diabetes, cardiovascular disease, arthritis, and cancer add to the number of patients being managed by healthcare providers, and to the ever-increasing amount of data generated during diagnosis and treatment.
- Increasing care management – Healthcare providers, health plans and employers have launched and expanded programs to work with patients, members and employees to help them proactively active their health and care. Monitoring and communication of health information is critical to the success of these programs.
- Increasing patient engagement – The last decade has seen shifts toward more “patient-centric” healthcare delivery models and demonstrated the benefits of increasing patient involvement with self-monitoring and participation in their own treatment.
- Increasing digitization – The evolution of high-speed networks and the increasing prevalence of smart phones, tablets, and mobile platforms in healthcare settings has been accompanied by government initiatives to promote digital healthcare platforms. At the same time, low doctor-to-patient ratios in many areas has led to an increased dependency on self-operated eHealth platforms.1
- New patient safety initiatives – Digitizing health data provides the opportunity for improved tracking, monitoring, and other Big Data initiatives to tighten medical and safety oversight and to identify opportunities for enhanced patient care.
Medical Display Device Landscape
The array of medical display devices in use today is extensive, ranging from the expensive medical-grade displays that are used for high-resolution diagnostic imaging within hospital and clinic settings, to the simple digital-read thermometers available from any drug store. Requirements for the various displays integrated into these devices can vary widely, for example “small, low-resolution panels (or even passive segmented displays) can be used in rugged and mobile medical applications.”4
Many medical devices—such as implantable pacemakers—do not incorporate displays for the obvious reason that they are placed inside the body where they are unseen by patients and providers. Some newer devices combine a sensor that is worn on the body or held that connects to an app on the patient’s smartphone without needing an integrated display screen. Devices that do typically incorporate embedded display panels include:
For an in-depth look at medical-grade displays, refer to Radiant’s 2019 blog post: “Medical Displays: New Technologies and Strict Standards Drive a Need for Visual Quality Testing”.
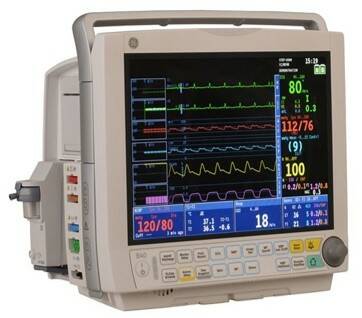
A multi-parameter patient monitor, which can simultaneously display a range of cardiac, hemodynamic, respiratory, pulse oximetry and neurology data, (Image: GE Healthcare), offering rugged performance and enhanced networking/connectivity.
Keeping Up With the Device Market
Medical devices have numerous quality and safety requirements they must meet, including (in the U.S.):
- ISO quality system requirements
- Complaint management and corrective action preventive action (CAPA)
- FDA 21 CFR Part 820
- FDA 21 CFR Part 11
- ISO 13485
Many consumer-use devices such as home monitors are considered “Class 1” medical devices according to the FDA—that is, they present “low to moderate risk” to patient health and safety.5 Class 1 devices require less oversight, but still must be registered as medical devices.
However, for medical device makers, “regulatory requirements are just one component—but incorporating emerging and continually evolving technologies requires device companies to raise their game if they want to stay ahead of the competition and leverage cutting edge technologies—from the Internet of Things (IoT) to artificial intelligence and machine learning to augmented reality and even robotics.”2 Displays are among the “continually evolving technologies” that go into this medical device mix.
As consumers have become accustomed to features like touch screens and apps in their every-day devices like cell phones and smartwatches, likewise they expect to find the same convenience and features in the latest medical devices. It can be a challenge for manufacturers to provide these display features while also meeting the high level of regulatory requirements that apply to medical use devices.
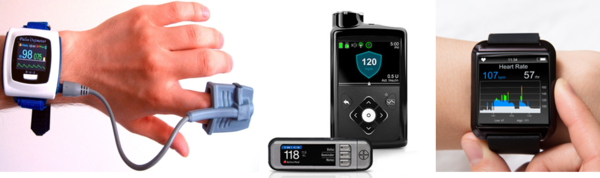
A wearable pulse oximeter (left), an insulin pump system for patients with diabetes (center), and a smartwatch heart rate monitor (right). (Left image: Wearable Medical Devices Market Research Report by marketdataforecast.com, center image: MiniMed)
For example, in this case study, Unisystem Displays outlines the extensive process they undertook to ensure the performance of a medical device with a TFT (thin film transistor) display and touch screen capabilities. They needed to perform exhaustive testing and troubleshooting to ensure the device could meet stringent electromagnetic compatibility standards before it could be approved for sale on the European market.
Medical Display Quality Considerations
As the broader display industry has developed new technologies such as LED, OLED, and microLED, the medical display device industry has evolved along a similar adoption path, albeit more slowly. New technologies must first prove their ability to offer both the luminance and rugged performance requirements of medical displays.
Medical device displays “can be subjected to extreme temperature, shock and vibration, moisture and variable ambient lighting. Those conditions mandate a highly reliable display and possibly the use of a clear protective cover that can safeguard the display from the rigors of daily use. Variable ambient-lighting conditions range from low-level indoor lighting to direct sunlight, in which case peak luminance and high contrast become very important.”4 Medical displays in a hospital environment can be in use 24 hours a day x 7 days a week x 365 days a year, subjected to moisture exposure from being wiped down for sanitation, and frequent bumps and vibration from being moved room to room throughout the facility.
Likewise, emergency respiratory devices are often used during medical transportation (such as in an ambulance or medevac helicopter), “which is one of the harshest operating environments in health care. The equipment will be exposed to climates ranging from Fairbanks, Alaska, to Tucson, Arizona. The new temperature range for numeric, graphic, and character LCDs in portable devices for hostile operating environments is -40°C to +85°C.”6
Devices for use in these environments typically don’t need high resolution, high color fidelity, or fast response times. “The demands are relatively simple because the information to be displayed is simple—maybe several scanned wave-forms and some numerical content. Even so, the displays need to be power-efficient and mechanically robust, with high reliability, high quality, and a small footprint.”4
For device displays used in some other settings or for specific functions, however, high-resolution image quality and visual performance is important. Two of the industry’s standards organizations have outlined extensive medical display performance parameters: the European DIN (Deutsches Institute for Normung eV) body and the American Association of Physicists in Medicine (AAMP). These organizations have specified acceptable performance for criteria including resolution, image noise, glare, color uniformity, geometric distortion, reflection, luminance response, luminance uniformity, and viewing angle.
In its most recent report on Display Quality Assurance in January 2019, the AAMP Task Force outlined its latest recommendations. While much of the report focuses on high-resolution “medical-grade” displays that are used for radiology and other diagnostic imaging purposes, it also addresses other types of displays used in clinical settings, grouped under the umbrella term “Electronic Health Record Displays.” So far, there does not seem to be a push for such stringent display standards for many of the medical wearables and consumer-use devices on the market, beyond general quality and performance considerations for device safety and accuracy.
Medical device makers—and the display module suppliers they often rely on—are still using LCD screens for a majority of uses. In part, this is due to a reluctance to submit new devices through the approval/registration process if a current LCD-based product exists. However, the brightness and performance of OLED displays offer advantages for certain medical device applications, such as glucometers and thermometers.4 Industry analysts are also predicting that microLED display technology will come to dominate the wearables sector7.
Testing Display Quality
For those device displays where luminance, uniformity, color, distortion, and other visual characteristics do have to meet specific standards—or where consumer expectations demand a high-quality visual experience—Radiant Vision Systems provides industry-leading display metrology solutions. Radiant’s ProMetric® Imaging Photometers and Colorimeters, in combination with our TrueTest™ Automated Visual Inspection Software, allow display manufactures to measure characteristics including luminance, chromaticity, contrast, and distortion, as well as to identify non-uniformity and defects like lines and spots (mura), particles, and stuck-on/off pixels.
To measure view angle performance, Radiant’s FPD Conoscope Lens enables high-resolution photopic measurement of the angular distribution of color, luminance, and contrast of a display without rotating the camera or display device. The lens uses Fourier optics to qualify display output as seen from +/- 70 degrees in a single measurement.

Radiant's ProMetric® Imaging Colorimeter and Photometer (left), TrueTest™ Software (center) and FPD Conoscope Lens mounted to a ProMetric I-Series Imaging Colorimeter (right).
CITATIONS
- "IoT Medical Devices Market by Product (Blood Pressure Monitor, Glucometer, Cardiac Monitor, Pulse Oximeter, Infusion Pump), Type (Wearable, Implantable Device), Connectivity Technology (Bluetooth, Wifi), End User (Hospital) - Global Forecast to 2023", report from MarketsandMarkets, August 2018
- Reedy, S., “A Pulse on Quality 4.0 for Medical Device Manufacturing”, Quality Magazine, December 10, 2019
- Slachta, A., “Medical wearables market to surpass $26 billion by 2026”, Cardiovascular Business, April 24, 2019, based on report from Transparency Market Research
- “Displays for Medical Applications”, New Vision Display, May 3, 2017
- “Classify Your Medical Device”, Food and Drug Administration, 8/31/2018 (Retrieved 1/14/20)
- “Designing Portable Medical Devices”, December 14, 2009, Newark.com (Retrieved 1/14/20)
- Chen, Y., "How Micro LED Supports Smart Wearable Applications", LEDInside, Decembe 2, 2019
Join Mailing List
Stay up to date on our latest products, blog content, and events.
Join our Mailing List
